20 Dec 2020 501 contributor considers whether or not he received the COVID-19 vaccine
By Mark McDonald
When I saw a news story about stage three trials for the Moderna vaccine, I wanted to know more. I registered my interest online and received an interview call within a few weeks. I had questions about safety, process, timelines and more. I learned that it was a “double-blind” trial, so I would not learn whether I had the vaccine or a placebo. Little did I know, that would be the most uncomfortable part of this trial.
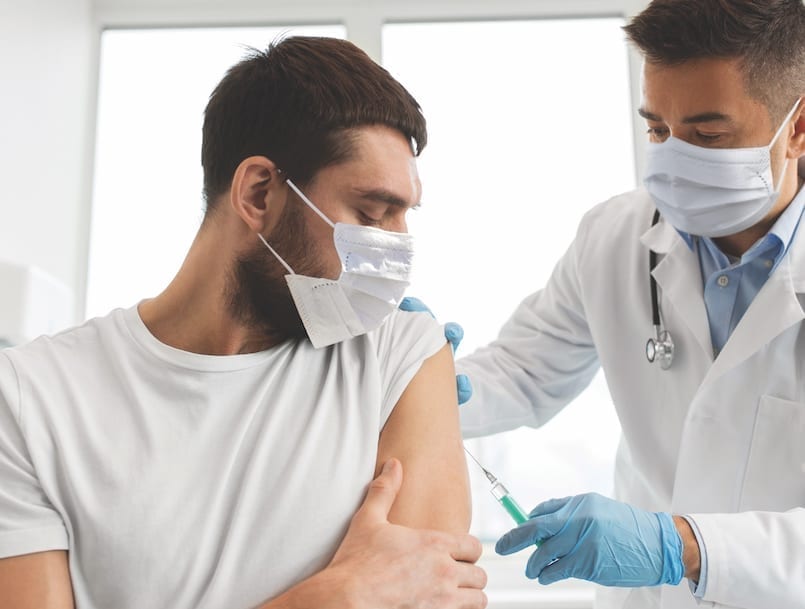
I was scheduled for tests to determine whether my health qualified for participation and completed an extensive questionnaire. I had read what I could find online about clinical trials and thought I knew what to expect. After the exam, I had a COVID-19 test and got ready to leave. Then, they told me that I could get my first vaccine that day because the study was allowed to move faster than expected. While I was confident that the trial was safe, that caught me off guard. Then I understood the name “Operation Warp Speed.”
I had the first vaccine and sat in observation for an hour. I enjoyed reading through the provided materials which included instructions should I need to visit an ER, a special number for questions or concerns, and more information about the process and expectations. After about an hour with no reactions, I figured I had gotten the placebo. My arm wasn’t even sore like it usually was with a flu shot. I celebrated with a nice lunch and then drove home, poking my arm to see if there was any soreness. “Must have been a placebo,” I thought.
That night, I had an upset stomach most of the night. My temperature never went up any significant amount, but my arm was sore the next day. Within 18 hours, I was back to normal and hopeful that I was safe from the virus. I had daily check-ins via an App, along with weekly calls and follow-up office visits scheduled. My questions mounted: “Was it real or a placebo?” Just in case, I followed the “three W’s” — I wore my mask, washed my hands and watched my distance.
My second shot visit was like the first, but without the reading materials. I shared about my upset stomach and soreness, and one of the nurses said she had not heard of anyone who had an upset stomach. That made me doubt even more if I had received the actual vaccine and I tried to remember what I ate for lunch that day. It all seemed to be going too easy to be the real vaccine.
After the second shot, my symptoms were again delayed until that evening. I had a slight fever, more soreness, and general achiness and fatigue for about 24 hours. I got back to normal, completed my daily questionnaire through their app, and answered weekly phone calls to check-in. And I continued to observe the “three W’s.”
I’ve run into several people I know who were part of the study, too. The discussion always followed the same script: “What was it like for you?” and, “Do you think you got the real thing?” One person said she had a fever and almost went to the ER because of her symptoms. But she also returned to normal after 48 hours and wondered if she received the real vaccine or if her symptoms were from something else.
Being in a double-blind study turns out to cause a roller coaster of questions and emotions. Some have thanked me for my willingness to risk my life, but I never felt that way. I’ve just learned to live with uncertainty, which is what we’ve all had to learn!
It has made me keenly aware of the risks every single one of us is taking, and how important it is for everyone to rise to our obligation — our duty — to watch out for each other. I have known dozens of people who have had to isolate and a few who tested positive. I’ve lost one friend and another colleague, and I’ve visited with dozens who have struggled deeply because of this pandemic. Being part of a trial seems relatively easy compared to what some have gone through … whether I got the real vaccine or a placebo.
Don’t be afraid of a shot that is based on years of research based on SARS and mRNA, especially since those of us who were in the study went through much of the same uncertainty as those who didn’t join the study.
Be safe, my friends.
- The beginning of the Christmas season - December 3, 2023
- Faith at the center of it all - February 28, 2023
- The spiritual connections between the Maker and makers - August 31, 2022











